1.7.3
Properties of Water
Test your knowledge with free interactive questions on Seneca — used by over 10 million students.
Useful Properties of Water
Water is a major component of cells and is essential to life as we know it (60–70% of the human body is made up of water). The properties of water that make it such a useful substance are:

Good metabolite
- Water is used or formed in many metabolic reactions, such as condensation and hydrolysis reactions.
- ATP + H2O → ADP + Pi + energy
- A bond is broken and a water molecule is used up and so this is a hydrolysis reaction.
- ADP + Pi + energy → ATP + H2O
- A new bond is formed and a water molecule is released and so this is a condensation reaction.
- ATP + H2O → ADP + Pi + energy

High heat of vaporisation
- The specific latent heat of vaporisation is the amount of energy needed to change 1 kg of a liquid substance to a gas.
- As liquid water heats up, hydrogen bonding makes it difficult to separate the water molecules from each other. This means that a lot of energy is needed for water to evaporate.
- When water evaporates, energy is used up - this cools the environment where the evaporation is taking place.
- This is why sweating helps with body temperature regulation.

High heat capacity
- Specific heat capacity is the amount of heat one gram of a substance must absorb or lose to change its temperature by 1oC.
- Water has a high heat capacity so takes a long time to heat and cool.
- The specific heat capacity of water is much larger than sand. This is why land cools faster than the sea.
- Water is used by warm blooded animals to more evenly disperse heat in their bodies.
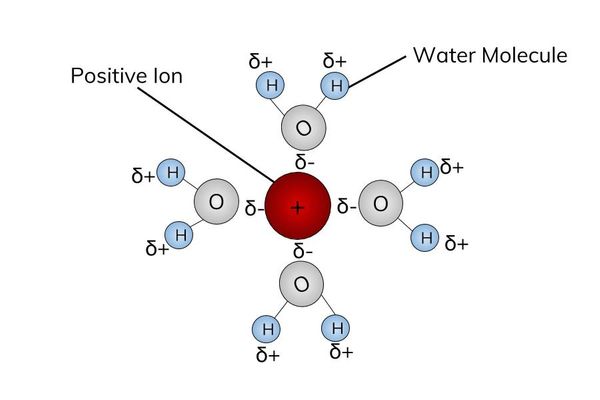
Good solvent
- Water is a good solvent because ions and polar molecules can easily dissolve in it.
- Water is a polar molecule. This means that the positive end of the water molecule attracts negative ions and the negative end will attract positive ions.
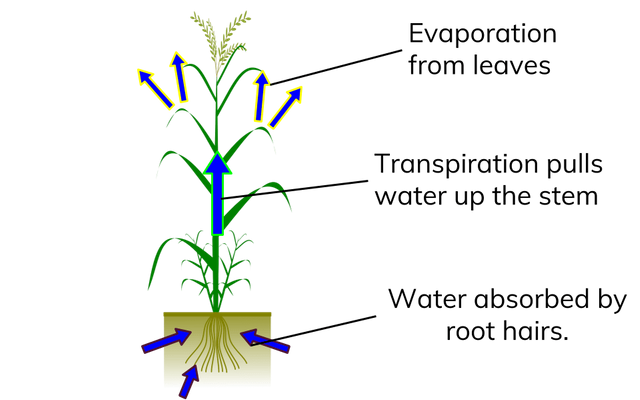
Cohesive properties
- The strong attraction between water molecules due to hydrogen bonds is called cohesion.
- Cohesion produces surface tension where water meets air.
- This is why water forms droplets when placed on a dry surface rather than being flattened out by gravity.
- Plants use this natural phenomenon to help transport water from their roots to their leaves.
1Biological Molecules
1.1Monomers & Polymers
1.2Carbohydrates
1.3Lipids
1.4Proteins
1.4.1The Peptide Chain1.4.2Investigating Proteins1.4.3Primary & Secondary Protein Structure1.4.4Tertiary & Quaternary Protein Structure1.4.5Enzymes1.4.6Factors Affecting Enzyme Activity1.4.7Enzyme-Controlled Reactions1.4.8End of Topic Test - Lipids & Proteins1.4.9A-A* (AO3/4) - Enzymes1.4.10A-A* (AO3/4) - Proteins1.4.11Diagnostic Misconceptions - Enzyme Inhibitors
1.5Nucleic Acids
1.6ATP
1.7Water
1.8Inorganic Ions
2Cells
2.1Cell Structure
2.2Mitosis & Cancer
2.3Transport Across Cell Membrane
2.4Cell Recognition & the Immune System
2.4.1Immune System2.4.2Phagocytosis2.4.3T Lymphocytes2.4.4B Lymphocytes2.4.5Antibodies2.4.6Primary & Secondary Response2.4.7Vaccines2.4.8HIV2.4.9Ethical Issues2.4.10End of Topic Test - Immune System2.4.11Exam-Style Question - Immune System2.4.12A-A* (AO3/4) - Immune System2.4.13Diagnostic Misconceptions - Humoral vs Cellular
3Substance Exchange
3.1Surface Area to Volume Ratio
3.2Gas Exchange
3.3Digestion & Absorption
3.4Mass Transport
3.4.1Haemoglobin3.4.2Oxygen Transport3.4.3The Circulatory System3.4.4The Heart3.4.5Blood Vessels3.4.6Cardiovascular Disease3.4.7Heart Dissection3.4.8Xylem3.4.9Phloem3.4.10Investigating Plant Transport3.4.11End of Topic Test - Mass Transport3.4.12A-A* (AO3/4) - Mass Transport3.4.13Diagnostic Misconceptions - Concentration Gradient3.4.14Diagnostic Misconceptions - Cardiac Cycle3.4.15Diagnostic Misconceptions - Carrying Capacity3.4.16Diagnostic Misconceptions - Translocation
4Genetic Information & Variation
4.1DNA, Genes & Chromosomes
4.2DNA & Protein Synthesis
4.3Mutations & Meiosis
4.4Genetic Diversity & Adaptation
4.5Species & Taxonomy
4.6Biodiversity Within a Community
4.7Investigating Diversity
5Energy Transfers (A2 only)
5.1Photosynthesis
5.1.1Overview of Photosynthesis5.1.2Photoionisation of Chlorophyll5.1.3Production of ATP & Reduced NADP5.1.4Cyclic Photophosphorylation5.1.5Light-Independent Reaction5.1.6A-A* (AO3/4) - Photosynthesis Reactions5.1.7Limiting Factors5.1.8Photosynthesis Experiments5.1.9End of Topic Test - Photosynthesis5.1.10A-A* (AO3/4) - Photosynthesis
5.2Respiration
5.2.1Overview of Respiration5.2.2Anaerobic Respiration5.2.3A-A* (AO3/4) - Anaerobic Respiration5.2.4The Link Reaction5.2.5The Krebs Cycle5.2.6Oxidative Phosphorylation5.2.7Respiration Experiments5.2.8End of Topic Test - Respiration5.2.9A-A* (AO3/4) - Respiration5.2.10Diagnostic Misconceptions - Aerobic vs Anaerobic
5.3Energy & Ecosystems
6Responding to Change (A2 only)
6.1Nervous Communication
6.2Nervous Coordination
6.3Muscle Contraction
6.4Homeostasis
6.4.1Overview of Homeostasis6.4.2Blood Glucose Concentration6.4.3Controlling Blood Glucose Concentration6.4.4End of Topic Test - Blood Glucose6.4.5Primary & Secondary Messengers6.4.6Diabetes Mellitus6.4.7Measuring Glucose Concentration6.4.8Osmoregulation6.4.9Controlling Blood Water Potential6.4.10ADH6.4.11End of Topic Test - Diabetes & Osmoregulation6.4.12A-A* (AO3/4) - Homeostasis6.4.13Diagnostic Misconceptions - Effect of ADH
7Genetics & Ecosystems (A2 only)
7.1Genetics
7.2Populations
7.3Evolution
7.3.1Variation7.3.2Natural Selection & Evolution7.3.3End of Topic Test - Populations & Evolution7.3.4Types of Selection7.3.5Types of Selection Summary7.3.6Overview of Speciation7.3.7Causes of Speciation7.3.8Diversity7.3.9End of Topic Test - Selection & Speciation7.3.10A-A* (AO3/4) - Populations & Evolution7.3.11Diagnostic Misconceptions - Types of Speciation
8The Control of Gene Expression (A2 only)
8.1Mutation
8.2Gene Expression
8.2.1Stem Cells8.2.2Stem Cells in Disease8.2.3End of Topic Test - Mutation & Gene Epression8.2.4A-A* (AO3/4) - Mutation & Stem Cells8.2.5Regulating Transcription8.2.6Epigenetics8.2.7Epigenetics & Disease8.2.8Regulating Translation8.2.9Experimental Data8.2.10End of Topic Test - Transcription & Translation8.2.11Tumours8.2.12Correlations & Causes8.2.13Prevention & Treatment8.2.14End of Topic Test - Cancer8.2.15A-A* (AO3/4) - Gene Expression & Cancer
8.3Genome Projects
Jump to other topics
1Biological Molecules
1.1Monomers & Polymers
1.2Carbohydrates
1.3Lipids
1.4Proteins
1.4.1The Peptide Chain1.4.2Investigating Proteins1.4.3Primary & Secondary Protein Structure1.4.4Tertiary & Quaternary Protein Structure1.4.5Enzymes1.4.6Factors Affecting Enzyme Activity1.4.7Enzyme-Controlled Reactions1.4.8End of Topic Test - Lipids & Proteins1.4.9A-A* (AO3/4) - Enzymes1.4.10A-A* (AO3/4) - Proteins1.4.11Diagnostic Misconceptions - Enzyme Inhibitors
1.5Nucleic Acids
1.6ATP
1.7Water
1.8Inorganic Ions
2Cells
2.1Cell Structure
2.2Mitosis & Cancer
2.3Transport Across Cell Membrane
2.4Cell Recognition & the Immune System
2.4.1Immune System2.4.2Phagocytosis2.4.3T Lymphocytes2.4.4B Lymphocytes2.4.5Antibodies2.4.6Primary & Secondary Response2.4.7Vaccines2.4.8HIV2.4.9Ethical Issues2.4.10End of Topic Test - Immune System2.4.11Exam-Style Question - Immune System2.4.12A-A* (AO3/4) - Immune System2.4.13Diagnostic Misconceptions - Humoral vs Cellular
3Substance Exchange
3.1Surface Area to Volume Ratio
3.2Gas Exchange
3.3Digestion & Absorption
3.4Mass Transport
3.4.1Haemoglobin3.4.2Oxygen Transport3.4.3The Circulatory System3.4.4The Heart3.4.5Blood Vessels3.4.6Cardiovascular Disease3.4.7Heart Dissection3.4.8Xylem3.4.9Phloem3.4.10Investigating Plant Transport3.4.11End of Topic Test - Mass Transport3.4.12A-A* (AO3/4) - Mass Transport3.4.13Diagnostic Misconceptions - Concentration Gradient3.4.14Diagnostic Misconceptions - Cardiac Cycle3.4.15Diagnostic Misconceptions - Carrying Capacity3.4.16Diagnostic Misconceptions - Translocation
4Genetic Information & Variation
4.1DNA, Genes & Chromosomes
4.2DNA & Protein Synthesis
4.3Mutations & Meiosis
4.4Genetic Diversity & Adaptation
4.5Species & Taxonomy
4.6Biodiversity Within a Community
4.7Investigating Diversity
5Energy Transfers (A2 only)
5.1Photosynthesis
5.1.1Overview of Photosynthesis5.1.2Photoionisation of Chlorophyll5.1.3Production of ATP & Reduced NADP5.1.4Cyclic Photophosphorylation5.1.5Light-Independent Reaction5.1.6A-A* (AO3/4) - Photosynthesis Reactions5.1.7Limiting Factors5.1.8Photosynthesis Experiments5.1.9End of Topic Test - Photosynthesis5.1.10A-A* (AO3/4) - Photosynthesis
5.2Respiration
5.2.1Overview of Respiration5.2.2Anaerobic Respiration5.2.3A-A* (AO3/4) - Anaerobic Respiration5.2.4The Link Reaction5.2.5The Krebs Cycle5.2.6Oxidative Phosphorylation5.2.7Respiration Experiments5.2.8End of Topic Test - Respiration5.2.9A-A* (AO3/4) - Respiration5.2.10Diagnostic Misconceptions - Aerobic vs Anaerobic
5.3Energy & Ecosystems
6Responding to Change (A2 only)
6.1Nervous Communication
6.2Nervous Coordination
6.3Muscle Contraction
6.4Homeostasis
6.4.1Overview of Homeostasis6.4.2Blood Glucose Concentration6.4.3Controlling Blood Glucose Concentration6.4.4End of Topic Test - Blood Glucose6.4.5Primary & Secondary Messengers6.4.6Diabetes Mellitus6.4.7Measuring Glucose Concentration6.4.8Osmoregulation6.4.9Controlling Blood Water Potential6.4.10ADH6.4.11End of Topic Test - Diabetes & Osmoregulation6.4.12A-A* (AO3/4) - Homeostasis6.4.13Diagnostic Misconceptions - Effect of ADH
7Genetics & Ecosystems (A2 only)
7.1Genetics
7.2Populations
7.3Evolution
7.3.1Variation7.3.2Natural Selection & Evolution7.3.3End of Topic Test - Populations & Evolution7.3.4Types of Selection7.3.5Types of Selection Summary7.3.6Overview of Speciation7.3.7Causes of Speciation7.3.8Diversity7.3.9End of Topic Test - Selection & Speciation7.3.10A-A* (AO3/4) - Populations & Evolution7.3.11Diagnostic Misconceptions - Types of Speciation
8The Control of Gene Expression (A2 only)
8.1Mutation
8.2Gene Expression
8.2.1Stem Cells8.2.2Stem Cells in Disease8.2.3End of Topic Test - Mutation & Gene Epression8.2.4A-A* (AO3/4) - Mutation & Stem Cells8.2.5Regulating Transcription8.2.6Epigenetics8.2.7Epigenetics & Disease8.2.8Regulating Translation8.2.9Experimental Data8.2.10End of Topic Test - Transcription & Translation8.2.11Tumours8.2.12Correlations & Causes8.2.13Prevention & Treatment8.2.14End of Topic Test - Cancer8.2.15A-A* (AO3/4) - Gene Expression & Cancer
8.3Genome Projects
Practice questions on Properties of Water
Can you answer these? Test yourself with free interactive practice on Seneca — used by over 10 million students.
- 1Water’s high heat capacity is caused by what? Multiple choice
- 2What % of the human body is made up of water?Multiple choice
Unlock your full potential with Seneca Premium
Unlimited access to 10,000+ open-ended exam questions
Mini-mock exams based on your study history
Unlock 800+ premium courses & e-books